Very Low and Standard Risk Favorable Histology (FH) Wilms Tumor.
The COG protocol AREN0532, was a research protocol designed for patients with very low and standard risk favorable histology (FH) Wilms Tumor. This study is now closed.
Patients eligible for this study were:
- Up to 30 years old
- Had Stage I-III FH Wilms tumor
Patients were stratified by clinical and biological risk factors.
Very low risk
Treated with surgery and observation alone:
- Stage I FH Wilms tumor
- Tumor weighs less than 550 g
- Children less than 2 years of age
The COG protocol AREN0532 found most patients meeting very low-risk criteria can be safely managed by nephrectomy alone with resultant reduced exposure to chemotherapy. Children who relapsed were more successfully retreated than prior stage I/FH children, probably because they were naïve to both radiation therapy and chemotherapy.
Standard Risk
Patients given dactinomycin, doxorubicin and vincristine and were NOT given radiation therapy (RT):
- Stage I, FH, Wilms tumors with loss of heterozygosity for 1p and 16q, AND either age more than 2 years or tumor weight more than 550 g.
- Stage II, FH, Wilms tumor with any weight of tumor or any patient age
Patients given dactinomycin, doxorubicin and vincristine PLUS RT:
- If they have no loss of heterozygosity for 1p and 16q and have Stage III, FH Wilms tumor.
- All Stage I and II patients with spill (including local needle biopsy) were reclassified as stage III based on NWTS 5 data showing inferior relapse free survival (RFS) for Stage II patients (4yr RFS 70% with spill versus 84% without spill).
Aim of study was to achieve the same excellent overall survival from previous studies, while continuing to reduce toxicity.
RT started at Day 1 for patients whose primary tumors were resected initially. For patients
with delayed tumor resection, RT started after the primary tumor was resected, usually
at Week 7 or 13.
The indications for RT were:
- Stage III FH Wilms tumor
- Relapsed Stage I FH Wilms tumor (all relapsed patients with pulmonary or intra abdominal tumor bed recurrence).
Indications for RT:
Site |
Indications & RT Dose |
Flank RT |
Stage III - 1080 cGy in 6#
Stage III local tumor spillage 1080 cGy in 6#
Flank or peritoneal biopsy, open biopsy, flank surgical spillage during surgery
Recurrent Wilms tumor - 1080 cGy in 6#
|
Whole Abdomen RT |
Stage III with:
Diffuse unresectable peritoneal implants - whole abdomen dose is - 21 Gy in 150 cGy fractions. Renal shielding needed to limit the dose to the normal kidney to less than 14.4 Gy.
Recurrent intraperitoneal Wilms tumor - 10.5 Gy at 150 cGy per fraction
|
Metachronous Wilms |
|
Lymph Node RT |
|
A boost of 1080 cGy was given to areas of residual tumor after surgery.
Flank RT
Treatment Volumes:
| Radiation Volume | Definition |
Gross Tumor Volume GTV |
|
Clinical Target Volume CTV |
|
Planning Target Volume PTV |
|
- The superior, inferior and lateral borders of RT field are placed at the edge of the PTV.
- The medial border of RT field extends across midline to include entire width of vertebral bodies (with a margin of 1 cm) at level concerned, but not so as to overlap any part of the contralateral kidney.
- RT field should not extend to the dome of the diaphragm unless tumor extends to that height.
- If positive lymph nodes have been surgically removed, then the entire length of the para-aortic chain of lymph nodes should be included in RT field
- An antero-posterior parallel-opposed technique (AP-PA) is recommended for flank RT.
Example of a flank field:
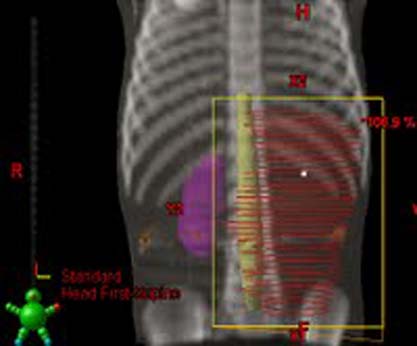
Whole Abdomen RT
The clinical target volume (CTV) is the entire peritoneal cavity
- Extending from the dome of the diaphragm superiorly to the pelvic diaphragm inferiorly, and laterally from the right to the left lateral abdominal wall.
- Femoral heads should be shielded during whole abdominal RT.
- An antero-posterior parallel-opposed technique (AP-PA) is used.
- When the total dose delivered is 2100 cGy (for diffuse peritoneal implants), renal shielding should be used to limit the dose to the remaining kidney to not receive more than 1440 cGy.
Extent of fields for Whole Abdominal RT:
Superior limit |
1 cm above the dome of the diaphragm. |
Inferior limit |
The bottom of the obturator foramen |
Lateral border |
1 cm beyond the lateral abdominal wall. |

