Central Nervous System
Leukoencephalopathy
Leukoencephalopathy is a broad term that means damage to the white matter of the brain. There are many different causes of this condition - including previous treatment for childhood brain tumors and leukemia.
Mulhern et al. evaluated quantitative MRIs (Q MRI) in young children given cranial RT (CRT) with and without chemotherapy for brain tumors10. There was a significant relationship between the amount of normal appearing white matter (NAWM) on Q MRI and age at CRT.
Loss of normal appearing white matter (NAWM) is associated with neurocognitive deficits and there is a direct correlation between the amount of loss of NAWM and the severity of the deficit11.
Etiology
Leukoencephalopathy develops as a consequence of treatment for acute lymphoblastic leukemia (ALL) with:
- Low dose cranial radiotherapy (RT)
- Intrathecal methotrexate (MTX)
This complication is most commonly seen after a combination of RT and MTX.
Methotrexate competitively inhibits the enzyme dihydrofolate reductase (DHFR) that is required for the synthesis of tetrahydrofolate, an important precursor for purine base synthesis. Therefore, it is thought that the pathophysiology of leukoencephalopathy is likely depletion of brain folate stores. In addition, increased concentration of homocysteine and excitotoxic neurotransmitters is also seen.
Signs and symptoms:
Leukoencephalopathy is characterized by white matter damage, but a prominent feature of the disease is memory loss and dementia. This suggests damage to gray matter.
Cognitive abnormalities likely reflect abnormalities of neurotransmission in the white matter.
The deficits associated with leukoencephalopathy may be mild to severe:
Mild |
Moderate |
Severe |
Mild memory loss with sequential memory problems
Attention deficits |
Marked deficits in attention
Memory problems
Visual spatial deficits
Fine motor incoordination
Executive function impairment
Poor math abilities
*verbal skills are less often affected |
*Children treated at a young age have greater and more global deficits
Difficulties with non-verbal tasks
Unable to process basic information
Severe attention deficit
Severe memory loss and dementia
Focal motor signs
Seizures
Ataxia
Death |
The neurocognitive decline is generally not evident until 3 years post-treatment and can be progressive for approximately 10 years after therapy.
Pathology:
- Demyelination
- Loss of oligodendroglia
- Focal or diffuse areas of white matter necrosis
- Mineralizing microangiopathy
- Dystrophic calcification
- Glial damage
Leukoencephalopathy generally spares the cortical gray matter and subcortical m fibres
Imaging:
Deep cerebral hemispheres and peri-ventricular areas most affected
CT scan shows:
- Calcification (especially of the basal ganglia)
- Hypodense areas
- Widened subarachnoid spaces
MR scan shows:
- Severity can be graded depending on radiological appearance of T2 MR scan
Table: Severity of leukoencephalopathy by MRI
Grade |
Appearance |
0 |
Normal |
1 |
Discontinuous area of periventricular hyperintensity (early - whitening in the periventricular region) |
2 |
Pencil thin, continuous line of hyperintensity around the ventricles |
3 |
Periventricular halo or band of hyperintensity of variable thickness forming smooth lateral margins around the ventricles |
4 |
Diffuse irregular white matter abnormality on T2 extending from the ventricles to the cortical medullary junction (late - diffuse coalescence of white and gray matter into an intense signal region and loss of structure). |
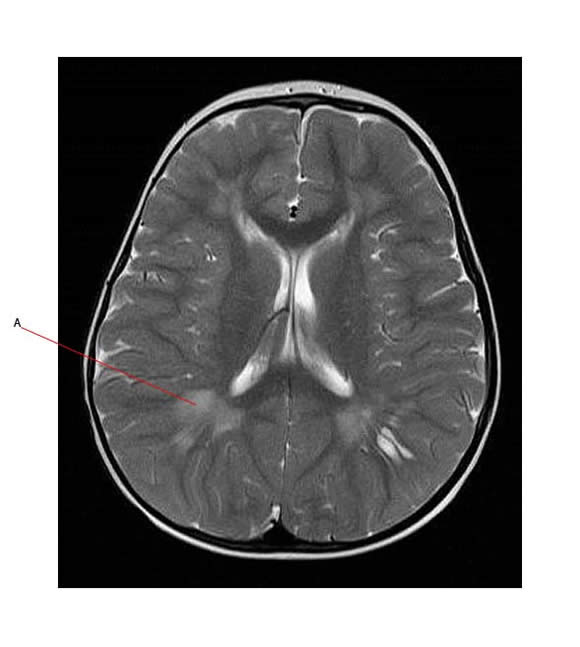
The MR below shows abnormality in white matter on T2 flair (A shows an area of abnormality)
Risk Factors:
Radiation Therapy (RT) -induced leukoencephalopathy is associated with:
- Total RT dose and fractionation schedule:
- Patients who receive higher doses of RT (>50 Gy) are more likely to develop leukoencephalopathy.
- Treatment volume:
- Whole brain RT is associated with higher grade leukoencephalopathy than focal RT.
- Time following RT:
- Patient age:
- Less mature brain structures also appear to be more sensitive to neurotoxic insult. Children younger than 5 years of age at diagnosis of ALL are more susceptible to long-term sequelae of treatment. This applies to both cranial RT and also CNS-directed chemotherapy
- Mulhern et al. evaluated young children given cranial RT (CRT) with and without chemotherapy for brain tumors. Young age at time of CRT was associated with worse scores on all IQ tests except verbal memory12,13
- Concurrent chemotherapy:
- Concurrent methotrexate or carboplatin therapy also increases risk.
- Retreatment (second course of RT)
- Retreatment with RT puts children at very high risk.
Methotrexate (MTX) -induced leukoencephalopathy:
- MTX is an important component of therapy for childhood leukemia and is given either IV or IT (intra-thecal) in ALL.
- MTX acts as an anti-folate that inhibits the formation of purine synthesis for DNA replication.
- Mechanism of MTX neurotoxicity is most likely multifactorial involving:
- alterations of folate metabolic pathways resulting in (subclinical) demyelination
- production of excitatory amino acids
- impairment of dopamine and serotonin synthesis.
Imaging in MTX associated leukoencephalopathy:
- Transient white matter changes can occur in children which then resolve after the end of therapy.
- Sometimes radiological abnormalities persist and these are not always associated with neurocognitive impairment (only if children are less than 5 years old and then the changes do correlate)
Girls more affected than boys
- Several studies have shown that girls perform more poorly than boys after therapy.
- This may be due to possible gender differences in brain maturation:
- an increase in white matter during childhood has been demonstrated to be smaller in girls than in boys, which could make girls more vulnerable to neurotoxic effects of chemotherapy.
- Girls tend to have more problems with visual spatial awareness, verbal IQ and alertness.
Risk factors for MTX-induced leukoencephalopathy
- Risk is increased if RT is given shortly after MTX treatment
- MTX-associated risks depends on:
- Cumulative amount
- Frequency of administration
- The absence of leukovorin in patients receiving IT MTX alone
- The use of triple intrathecal therapy
- Risk is increased if the leukemia has infiltrated the CNS
- This causes a delay in clearance of MTX from the ventricles and therefore, increases the level and duration of exposure

